
Advocating for Change
It's time to listen to the experts, to learn from the evidence, and to implement strategies to get things done — fast. The crisis is solvable. Change starts with you.
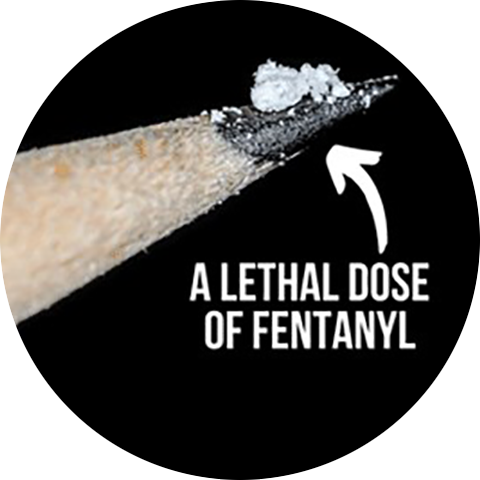
We're working to prevent fentanyl overdoses
See what we're doing to expand access to naloxone and fentanyl test strips, monitor overdose deaths, get funding for prevention programs, and increase public awareness.

President Joe Biden Signs Important SUD Legislation
On December 29, 2022, President Biden signed the Consolidated Appropriations Act into law. This Act includes three bills that will greatly improve treatment for substance use disorder.

You can help make change
Learn how to be an effective advocate in your home state.
Read our research

Medicaid & Collaborative Care
The Collaborative Care Model (CoCM) is an effective way to integrate mental health and substance use disorder treatment into primary care settings.

County Buprenorphine Access
Treatment with buprenorphine is highly effective. But our report shows county by county disparities and illustrates that over 1,200 counties in the U.S. lack access to buprenorphine.

The Fentanyl Epidemic
Fentanyl overdose deaths increased by 14% in 2018. This report outlines solutions to this growing national crisis to save countless lives.



